|
But the valency of elements, when combined with H or O first, increases from 1 to 4 and then it reduces to zero. While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8. Variation Of Oxidation State Along a Period. And atomic mass always increases in a period or a group. Periodic Trends in the Oxidation States of Elements. Note: Remember that the atomic masses which are mentioned in the periodic table of elements for different elements actually represent their average atomic masses. You can see that The molar mass of Potassium is 39.098 g/mol. So let’s look at the molar mass of Potassium, Phosphorus and Oxygen from the above periodic table. Now in K3PO4, there are 3 Potassium atoms, 1 Phosphorus atom and 4 Oxygen atoms. 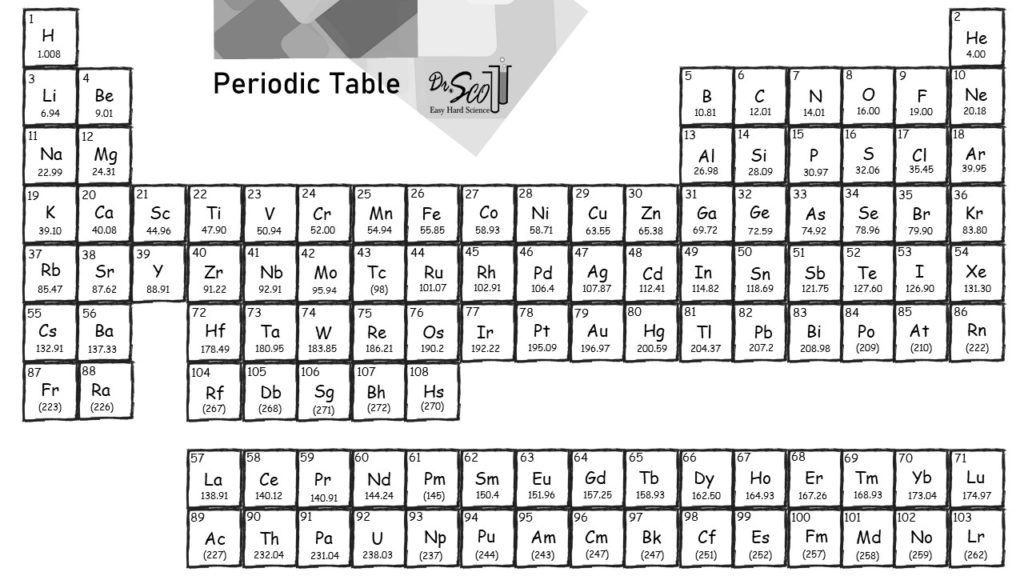
Thus, we can say that when we move down a group in case of non-metals, the reactivity of elements decreases while moving from left to right it increases in case of metals.Īlso, the atomic mass of the element actually uses average atomic masses of elements because naturally occurring elements exist as more than one isotopes which are the atoms of the same element which possess different relative masses. You can see the molar mass value of all the atoms from this periodic table. And the atomic mass on moving from left to right in the periodic table increases because the more protons and neutrons are added to the nuclei of an atom.Ītomic mass of an atom also increases while moving from top to bottom because the atoms in the rows, on further moving down, have more protons and nucleons. 1 The molar mass is a bulk, not molecular, property of a substance. 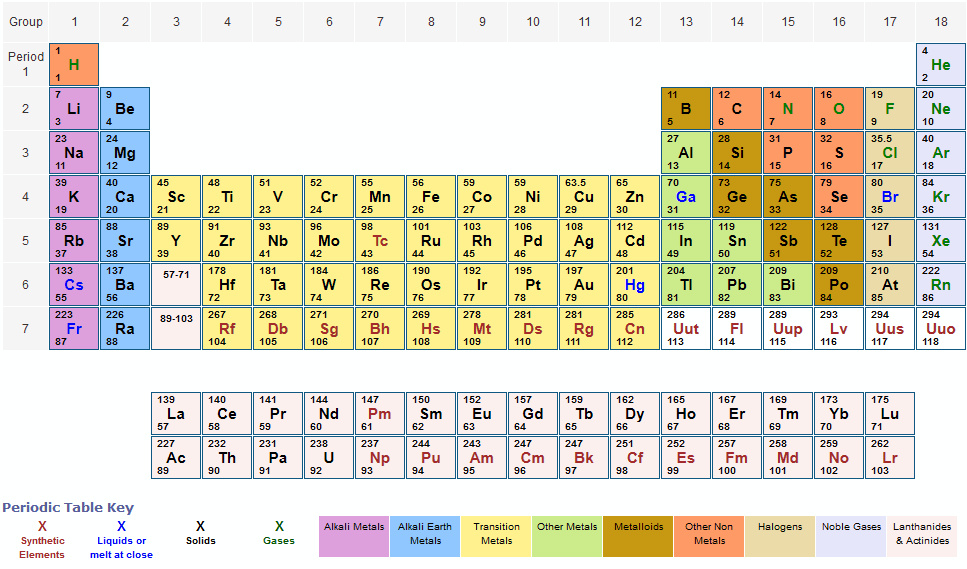
So, let us talk about one of the trends observed for the Atomic masses is defined as the sum of the total number of protons and neutrons of an atom. In chemistry, the molar mass ( M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. EA also demonstrates some periodic trends, although they are less obvious than the other periodic trends discussed previously. which increases or decreases down the group or while moving from left to bottom.Īs we know that a lot of trends are there in the physical and chemical properties of elements. So the molar mass of glucose is going to be six times the molar mass of carbon plus 12 times the molar mass of hydrogen plus six times the molar mass of oxygen. And so now we have all the information we need from our periodic table of elements. 
Now we interpret the coefficients as referring to molar amounts, not individual molecules.Hint: As we know that the periodic table contains a trend for every parameter like atomic number, electronegativity, ionisation enthalpy, electron gain enthalpy etc. Oxygen, we can see from our periodic table of elements, it has a molar mass of 16.00 grams per mole. We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as 
When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
